|
Carbon conducts electricity (but not as well as metals)Įlements that have both strong metallic and nonmetallic characteristics are metalloids.Carbon has the highest melting point of any element.The staircase inside the periodic table separates the from the. /periodic-table-58ea5e0a5f9b58ef7ed0a788.jpg)
They have the properties of both metals and nonmetals. Most of the elements on the border of this stair step are called metalloids. Explain that this separates the metals from the nonmetals. Nonmetals have exceptions too for example: Point out the bold stair step line on most periodic tables. Can form both anions (negative charge) and cations (positive charge).Brittle (opposite of ductile/malleable breaks instead of bending).The states of the elements is also indicated at room temperature of 25 o C. It can also be broadly divided into metals, nonmetals, and metalloids (semimetals). Properties of Nonmetals (opposite of metal properties) 1: The periodic table is split into seven periods (rows) with the inner transition metals shown separately on the bottom to decrease the width of the periodic table. Many metals are exceptions to these trends. Nonmetals are in fact more likely to gain electrons from other atoms instead. Elements with strong Coulombic forces holding their outer electrons close are less metallic, and they tend not to lose their outer electrons. These metallic elements have weaker Coulombic attractions between nucleus and outer electrons. On the periodic table, the elements colored yellow, which generally border the stair-step line, are considered to be metalloids. Students then use the color coded version of the table to answer. It is also good way to reinforce the metalloids and how they fall along the zig-zag line on the table. By color coding the elements it becomes very clear to students. shown in the periodic table, metals are the elements starting from Lithium with atomic number 3. Metalloids can also be called semimetals. As you can see from this color coded version of the Periodic Table, most of the elements are metals. This is a great way for students to visually see where on the table the metals tend to be versus the nonmetals. Elements that lose electrons easily are most metallic. A metalloid is an element that has properties that are intermediate between those of metals and nonmetals. Metallic Character comes from an element’s tendency to lose electrons and form positive ions (cations). Updated: 07-22-2021 Organic Chemistry I Workbook For Dummies Explore Book Buy On Amazon Using the periodic table, you can classify the elements in many ways. Some elements are hard to classify as metal, nonmetal, or metalloid for example, some chemists consider astatine (At) a nonmetal while others call it a metalloid. Metalloids are the six or so elements with properties of both metals and nonmetals. They lie along a staircase between the metals and nonmetals. Beginning at the top left of the table, or period 1, group 1, is a box containing “1 hydrogen H nonmetal gas and 1.008.” There is only one other element box in period 1, group 18, which contains “2 helium H e nonmetal gas and 4.003.” Period 2, group 1 contains “3 lithium L i metal solid and 6.As you can see from this color coded version of the Periodic Table, most of the elements are metals. Each element will be described in this order: atomic number name symbol whether it is a metal, metalloid, or nonmetal whether it is a solid, liquid, or gas and atomic mass. Metalloids are elements that look like metals and in some ways behave like metals but also have some nonmetallic properties. 
It is labeled “Symbol.” Below that is the number 1.008 which is labeled “Atomic Mass.” Below that is the word hydrogen which is labeled “name.” The color of the box indicates that it is a nonmetal. Just as shown in the table above, metals and nonmetals on the periodic table are often separated by a stairstep diagonal line, and several elements near this line are often called metalloids (Si, Ge, As, Sb, Te, and At). The number 1 is in its upper-left hand corner and is labeled “Atomic number.” The letter “H” is in the middle in red indicating that it is a gas. They are found between the metals and nonmetals. To the left of this box is an enlarged picture of the upper-left most box on the table. Above picture shows you the labeled Metalloids on the Periodic table. The 18 columns are labeled “Group” and the 7 rows are labeled “Period.” Below the table to the right is a box labeled “Color Code” with different colors for metals, metalloids, and nonmetals, as well as solids, liquids, and gases. 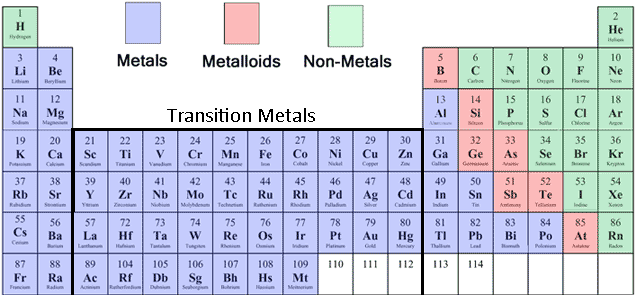
\): Elements in the periodic table are organized according to their properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
